Two deposition modes: dry deposits and wet deposits
In ordinary life, apart from the vicinity of the most serious accidents sites, their residual contamination and their impact are very low. Effects are mitigated by time. The most serious of these accidents, Chernobyl, is now more than a 30 years old. The time has not yet done its work in Fukushima for which ground deposits are less important. One should mention also the explosions of Khystim(1957) in the former Soviet Union and Windscale in the UK. These two former events of lesser magnitudes were related to the military programs of the time.
Radioactive deposits resulting from a major accident are sources of lasting ground contamination. When contamination exceeds permitted limits, the area should be decontaminated when possible, or people evacuate. This was the case at Chernobyl, where a circular area of 30 km radius has been emptied of its inhabitants. In the case of Fukushima, the population was evacuated in a radius of 20 km around the damaged plant, but it is too early to know how much of this area would be reoccupied.
Radioelements found in the atmosphere after a radioactive release settle to ground in two ways, called wet and dry.
Dry deposits occurs during the dispersal of a radioactive plume. The deposit is formed when the radioactive particles from the air enter in contact with the ground. It is particularly important when the radioactive atom concentration is high and air pollution lasts.
Wet deposits are due to rainfall or snowfall. Raindrops and snowflakes leach radioactive particles of the air. They bring them to the ground thereby resulting in a much more intense deposit than with dry deposition
With moisture, radioactive atoms penetrate into the soil leaving a residual contamination at the origin of an ambient external exposure. Part of the deposit remains where it is formed, but another part will trickle on the surface to join streams. Caused by rain, the transport runsoff down the slopes or on hard surfaces, such as roofs and roads, leading to uneven contamination distributions, with areas less affected and other much more.
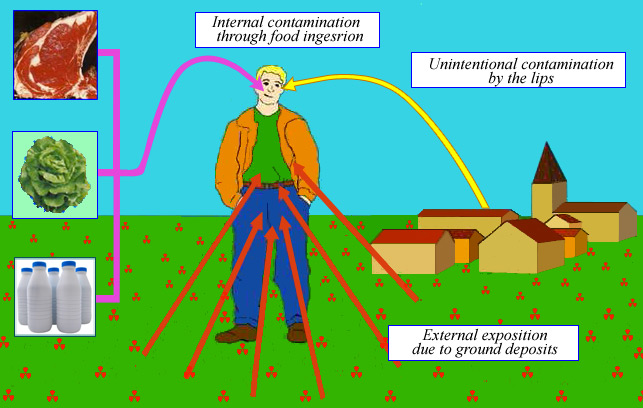
Radioactive deposits: Long term contamination
Ground deposits are long lasting. There is the risk from chronic exposure rate coming from ground and involuntary ingestion of radioactive particles (labile deposits). The main danger is that of an internal exposure resulting from ingestion of contaminated food. Radioactive atoms are first deposited on grass, leafy salads and vegetables. Later they are absorbed by the roots. Those deposited on the grass contaminate the fresh milk and meat of the cows grazing this grass.
© IRSN
Ground deposits : what dangers ?
Ground deposits are long lasting and present two dangers: the least is a chronic external exposure due to background gamma radiation; the most serious is the risk of internal exposure from ingestion of contaminated food from crops, mainly leafy vegetables and fresh milk
Iodine-131 is the source of the most dangerous contamination, a fortunately short-lived one because it disappears after three months. At the Chernobyl accident, the delay to take appropriate measures by the Soviet authorities, and to inform the population, was the origin of thyroid cancers among children and teenagers particularly by the consumption of contaminated milk. Now only remain essentially ground caesium deposits in the most contaminated areas. The most contaminated areas of Chernobyl, with caesium-137 surface activities exceeding 555 kBq / m2, are situated in Belarus ,over 7000 km2 representing 3.4% of its territory.
At Fukushima, caesium-137 is accompanied by caesium-134 whose radioactive half-life is 2 years and has not had time to decrease significantly. The deposits are concentrated of a strip of 20 km by 50 km northwest of the plant, In this 1000 km2 area, the combined activity of two isotopes exceeded 600 kBq / m2 in 2011.
Although a kBq (kilobecquerel) is a small unit, the activities of hundreds of kBq remain considerable (555 kBq represents the activity of a small source of 15 micro curies). Part of this ground radioactivity pass into plants. Foodstuffs from contaminated areas should be controlled and monitored. But measurements made by radioprotection agencies show that root uptakes is much less than that by the leaves uptakes happening in the first year and decreases over time.
In the long term, the main exposure is due to penetrating gamma rays from caesium-137. In the most contaminated areas around Fukushima, where a whole year exposure may exceed 50 millisieverts (mSv), the return of populations is not foreseen. In areas where the exposure would be below 20 mSv/year, residents were allowed to return.
Other articles on the subject « Accidental Releases »
Atomic bombs legacy
A legacy of the cold war The residue of radioactive contamination which still endures has its ori[...]
Atmospheric Releases
Atmospheric radioactive pollution after a major accident During a major accident, radioactive mat[...]
Risks of Internal Exposure
After an accident : what impact on food and man ? A large amount of radioactivity was dispersed i[...]
Exposition modes (accident)
What are the exposition pathways after a major accident ? What is the impact on man of a major re[...]