A generally transient exposure due to gamma rays
The majority of exposure to radiation is external – which is to say that the source of radioactivity is located outside the object or organism which is absorbing the rays. This is the case for natural radioactivity of cosmic radiation and the rays emitted by rock formations. The same also applies to medical exams, X-rays, and most accidental exposure.
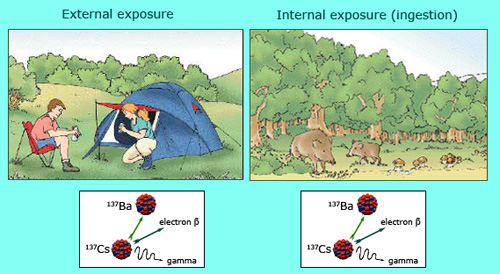
External and internal exposure to caesium-137
Deposits of caesium 137 coming from old atomic tests or the Chernobyl accident are responsible for both external and internal irradiations. Caesium 137, which emits gamma rays and beta electrons, contaminates layers of soil and plants. The campers on the left image are directly exposed to gamma rays emerging from the ground – gamma rays which are specific of caesium and easy to identify. As shown on the right, wild boars and mushrooms accumulate from the ground caesium, which later concentrate into the food chain. It is beta electrons which then become the primary source of internal irradiation. However, several months of camping and eating many portions of wild boar are needed before absorbing a smalp 1 millisievert dose.
© IRSN/dessin : Martine Beugin
When it comes to natural or accidental exposure, external radiations do not target a specific organ or area of the body. This is in direct contrast with internal exposure, where the radiation can target the bones, the thyroid gland, or any body part in close proximity to the source. External radiation can be made localised, however, as with medical or industrial scanning and tumour-specific therapy.
External exposure is usually caused by gamma rays, as neutron irradiations are extremely rare outside of nuclear power plants. Exposure to alpha rays, which can lead to burns on the skin, necessitates direct contact with the source as even a single sheet of paper is enough to stop the massive alpha particles. Beta radiation, which can also be felt over short distances, can likewise easily be stopped.
Gamma radiation causes the least damage of all natural radiation. This is due in part to the fact that many gamma rays pass straight through the irradiated bodies without interacting, and that the resultant effect spreads throughout the entire body.
Certain radioactive isotopes are responsible for both external and internal exposure. Caesium-137, emits an external characteristic gamma rays from deposits on the ground but emits also beta rays and can contaminate the food chain. For Iodine-131, a distinction has to be made between the irradiation of the thyroid gland caused by ingestion and the less harmful rays which come from iodine decays outside the body and do not target the thyroid gland.
External radiation only lasts for as long as the organism is exposed to the source. By opposition, if a radioactive substance is ingested, it can decay over months and even years within the body, emitting potentially harmful rays over the whole of this period.
External doses can be calculated from dosimeters sensitive to different types of radiation. By law, such dosimeters must be worn by all individuals working in a radiation environment: including nuclear industry workers, research and hospital workers. In radiation therapy, the dose absorbed by the tumour is carefully controlled by dosimeters placed on the skin and along the paths of all incoming rays.
Without dosimeters, it is impossible to measure the doses absorbed by individuals. This does not much matter when it comes to low doses, but in the case of severe exposure experts estimate exposures based on models such as at Hiroshima-Nagasaki and Chernobyl. Experts can only estimate average doses obtained from the data available. These averages, calculated after the event, rely on the hypotheses made, ignore what may have happened to each individual.