A radioactive and strategic element
The uranium atom is the heaviest atom present in the natural environment. Its radioactivity is very low. Its very long life of several billion years has allowed uranium to be still present. It is a rare chemical element found in the Earth’s crust with an average of 3 grams per tonne.
The uranium image has suffered from its association with the first atomic bombs. Its reputation as a malevolent radioisotope, however, is undeserved: in fact, the decay rate of uranium is among the slowest known to man. The activity of a sample of uranium could be compared to the water flow escaping from a pond through a pinprick.

Martin Heinrich Klaproth
Martin Heinrich Klaproth (1743-1817) was a German chemist best known for his discovery of a variety of chemical elements, including the 1789 discovery of uranium, zirconium and chromium. He is sometime credited as being the ‘father of analytical chemistry’.
© DR
These reassuring features not prevent this unfortunate element to be regularly presented by TV channels as a dangerous radioactive substance. Is our complacence born of ignorance? Contrary to the widespread fears, uranium presents low risks owing to its very low radioactivity. Its radioactive toxicity, according to experts from the CEA, is a hundred times weaker than its chemical toxicity, which itself is no different from the chemical danger posed by common heavy elements such as lead.

Uranium mine in Canada
Athabasca plateau in Saskatchewan province, Canada. Western Canada is particularly rich in uranium, with often between 28 to 210 kilograms of uranium per tonne as opposed to the usual 3 grams per tonne found elsewhere. This high abundance, taken in conjunction with difficult geological conditions and harsh climate have made the extraction process an almost entirely automated one.
© HARRY GRUYAERT /MAGNUM /AREVA
All isotopes of uranium are unstable and radioactive, but uranium 238 and uranium 235 have half-lives sufficiently long to have allowed them to still be present in the Solar System and on Earth. The half-life of uranium 238 is of 4.5 billion years, while uranium 235 has a half-life of ‘only’ 700 million years.
While both isotopes were at the time of Earth formation equally abundant, natural uranium today consists today of 99.3% uranium 238 and only 0.70% uranium 235. The nuclei of uranium 235 and 238 are, along with those of thorium 232, the heaviest present in nature. They were all formed billions of years ago by the explosion of heavy stars (supernovae).

Yellow cake
The radioactivity of uranium is low, and so no particularly high standards of radioprotection are needed: as can be seen with the above workmen. The concentrated uranium they are handling, also known as ‘Yellow Cake’, takes the form of a bright yellow powder containing about 750 kg of uranium per tonne. This high concentration makes it much easier to transport the uranium from the mine to the factory. The photograph above shows the ‘yellow cake’ on a filter at the Haute-Vienne Jouac treatment plant in Limousin, France.
© PHILIPPE LESAGE/AREVA
From a chemical point of view, uranium belongs to the actinide family, a group of atoms whose properties are similar to those of actinium, the 89th element in Mendeleyev’s periodic table. Uranium can be found in the Earth crust at 3 parts per million, particularly in granite and volcanic rocks. Certain uranium compounds (hexavalent ones) are highly soluble whereas others (tetravalent) are not.
As a particularly heavy element, uranium isotopes are primarily alpha emitters, though these radiations are sometimes accompanied by gamma rays.
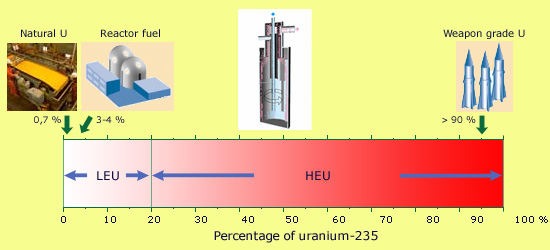
Weapon-grade and civilian uranium
Natural uranium is poor in the fissile isotope, containing as it does only 0.70% of uranium 235. It must be enriched before it can be used as a fuel in any commercial reactor. These reactors are powered by uranium which is enriched to have anywhere between 3 and 4% of uranium 235. In order to have an atomic bomb, the uranium has to be enriched to above 90%. The boundary between the uranium meant for civilian uses (LEU or low-enriched uranium) and that uranium meant for military use (HEU or highly-enriched uranium) is generally fixed at 20%.
© IN2P3
Uranium 235 is the only natural nucleus that can easily undergo fission. Highly sought-after, it can be used as a fuel in nuclear reactors and as an explosive in atomic bombs.
The more abundant uranium 238 is sometimes called fertile. Fission occurs comparatively rarely, and even under bombardment with energetic neutrons the probability of fission remains very low. What happens more frequently is that a neutron capture causes the nucleus to become unstable. After a few days, uranium 239 has transformed into plutonium 239, a radioisotope with a half-life of 24,000 years. Plutonium 239 is highly fissile and can also be used as a nuclear fuel or an atomic explosive.
Uranium reserves which exist under the oceans are about a thousand times more abundant than the reserves found in high quality minerals on land. Underwater uranium, however, would be extremely costly to exploit.
TO KNOW MORE ABOUT URANIUM AND ITS USES :
– 1) : Uranium isotopes
– 2) : Uranium fuels
Other articles on the subject « Main Radioactive Nuclei »
Plutonium 239
Plutonium 239: an artificial fissile nucleus, highly sought-after and feared Plutonium, the ninet[...]
Plutonium Properties
A transuranic element with long-lived radiotoxic isotopes Plutonium is a very dense metal, radioa[...]
Radium
The radioactive nucleus that made History Radium is an extremely rare element that was first disc[...]
Carbon-14
A by-product of cosmic rays The nucleus of carbon 14 contains 6 protons and 8 neutrons, instead o[...]
Potassium-40
A curiosity of Nature and a very long lived beta emitter Potassium 40 is a radioisotope found in [...]
Iodine 131
Radioactive iodine : A dangerous and short lived fission product Iodine 131 is a radioisotope wit[...]
Tritium
A radioactive isotope of hydrogen Tritium is a beta-emitting radioactive isotope of hydrogen. Its[...]
Caesium 137
A legacy of atmospheric nuclear bomb tests and accidents Caesium 137 is a radioactive element wit[...]
Strontium-90
A fission product with properties close to calcium Strontium-90 is with cesium-137 a major radioa[...]
Technetium 99
A pure gamma emitter widely used in nuclear medicine Of all the atoms below uranium in Mendeleyev[...]