Three naturally occurring isotopes, three produced in reactors
Natural uranium is constituted primarily of uranium-238 with 0.7% uranium-235 and a small amount of isotope 234. Three isotopes, uranium 236, 233 and 232, are also produced by reactors from uranium-235 and thorium. These three natural isotopes and these three artificial isotopes are alpha emitters
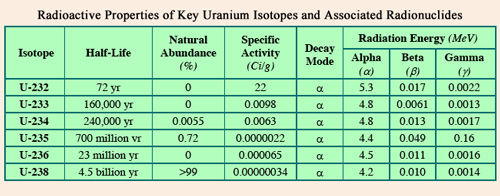
Comparison of radioactive properties of uranium isotopes
The main isotopes of uranium contained in this table have extremely long lifetimes with the exception of uranium 232. All are alpha emitters of 4 to 5 MeV of energy. The left columns of the table show however the presence of a low energy gamma radiation and rare decays beta. Specific activities (activities given for 1 gram) are inversely proportional to half-lives.
© ANL/Uranium fact sheet
Three naturally occurring isotopes
Uranium 238, which alone constitutes 99.3% of natural uranium has the longest lifetime: its period is 4.5 billion years, about the age of Earth. It is not very radioactive. Its very long period says it is still present in the Earth crust. Neutron capture by this nucleus leads to the formation of fissile plutonium-239 in a reactor. Hardly fissile, U-238 contributes to the operation of reactors and production of electricity through this plutonium. This impressive potential of fission energy remains still largely unexploited. The purpose of the fourth-generation breeder reactors is to recover this fantastic potential.
Uranium 235, the only existing fissile nucleus found in natural uranium, is used as a nuclear fuel in reactors and as an explosive for nuclear weapons. This very rare isotope, present at the concentration of 0.7% in natural uranium, is thus a highly strategic and coveted material. Its very long half-life (or period), 700 million years, is however, 6.5 times shorter than that of the isotope 238. At the time of the formation of Earth, U-235 was 85 times more abundant. The 0.7% observed today are a pale residue of this past abundance. If humans had been present at the beginning of Earth, they would not have needed to enrich uranium to make atomic bombs or operate their reactors!
Uranium 234 is the first long-lived descendant of uranium-238. In a natural sample of uranium, these nuclei are present in the unalterable proportions of the radioactive equilibrium of the uranium-238 filiation at a ratio of one atom of uranium-234 for 18 800 atoms of uranium- 238, so that the two isotopes contribute equally to the radiations emitted by uranium.
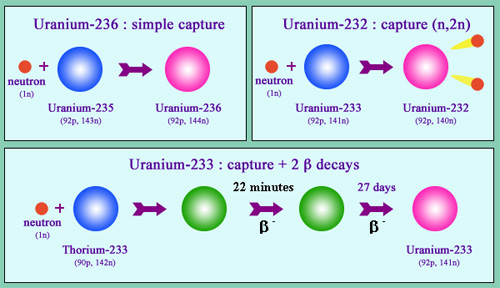
Formation of isotopes 236, 233 and 232
The isotopes 236, 233 and 232 of uranium are formed in reactors from captures of neutrons not followed by fission. Uranium-236 is formed by a simple radiative capture by a nucleus of uranium-235 non followed by fission. Uranium-233 is formed by a similar capture by a nucleus of natural thorium, followed by two radioactive decays. Uranium 233 is fissile itself. Submitted to the flow of neutrons from a reactor, it undergoes fission, but rarely turns into uranium 232 by a specific capture reaction (n, 2n) that triggers the expulsion of two neutrons.
© IN2P3
Three artificial isotopes
Uranium 236 is formed in the nuclear fuel from uranium 235, after neutrons captures that did not cause fission. The presence of this isotope in a sample of uranium means that the sample has beeninto a reactor.
Uranium 233 is a fissile nucleus that does not exist naturally, such as plutonium 239, which it is close to by its mode of production. It is produced by neutron capture in reactors containing thorium. Fissionable by fast neutrons and slow neutrons, this nucleus has some interesting features for energy production. Reactors using thorium and uranium-233 are one of the options considered for the future fourth-generation reactors.
Uranium 232 is a by-product of the reactors running with thorium and uranium 233 foels. The formation of this isotope results from specific neutron capture by uranium 233 that cause the ejection of two neutrons. Uranium 232 has a relatively short period of 68.9 years, but especially its radioactive filiation generates a descendant, thallium 208, that emits gamma rays of 2.6 MeV which are very energetic and highly penetrating. These intense radiations make handling of fissile uranium-233 contaminated with uranium 232, far more dangerous than conventional uranium 235 or plutonium 239 fuels. They constitutes an obstacle for the proliferation of bombs made of this fissile uranium.
Other articles on the subject « Nuclear Fuel »
Cycle Front End
From the extraction of uranium to the fuel fabrication The front-end of the nuclear cycle is the [...]
Isotopic Separation
The access key to the fuel of modern nuclear power plants … and to atomic weapons The uranium fue[...]
Ultracentrifugation
A separation process economical in energy, but proliferating The principle of centrifuges has lon[...]
Gaseous Diffusion
The first enrichment process, a large consumer of electricity The gaseous diffusion process was t[...]
PWR fuel assemblies
Fuel assemblies in the core of PWR Reactors The core of a pressurized water reactor (PWR) is cont[...]
Reactor fuel layout
Fuel assemblies in the core of nuclear reactors The nuclear fuel assemblies, that vary from one r[...]
Plutonium Use
A fissile element produced in nuclear reactors In February 1941 the American physicist Glenn Seab[...]
Pu : Fuel, bomb or Waste ?
Plutonium dark and bright sides … Sensitive material, like the roman god Janus, plutonium h[...]
MOX fuels
Introducing plutonium in nuclear fuels For energy production, plutonum is gold ! One gram of plut[...]
Thorium Fuels
Uranium 233: a fissile nucleus made from thorium Thorium is more abundant than uranium in the Ear[...]