Gamma can be attenuated but never fully stopped
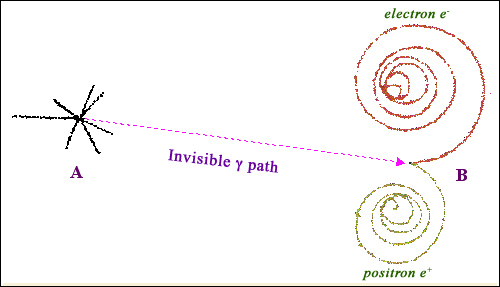
Trajectory of a gamma ray in a bubble chamber
Photons deposit their energy in matter by transferring it to charged particles. The bubble chamber picture above displays the birth, life and death of an individual gamma photon. The gamma was produced at point A, during a collision with a particle originating from an accelerator. After a certain distance travelled without any interaction, the gamma interacts at point B. It transfer its energy by creating an electron/positron pair. The presence of a very strong magnetic field causes the two oppositely-charged particles to spiral and gradually lose their energy before coming to a complete stop.
© IN2P3
The neutral gamma rays leave very different effects on their surroundings from those left by charged particles. Whereas alpha and beta rays transfer their energy to the environment by a gradual process, gamma photons transfer either everything or nothing. They have no effect until they interact with a nucleus or an electron, in which case they cause charged particles to move, thereby indirectly transferring their energy to the medium.
Gamma photons have three ways to interact with matter : photoelectric effect, Compton effect and pair production
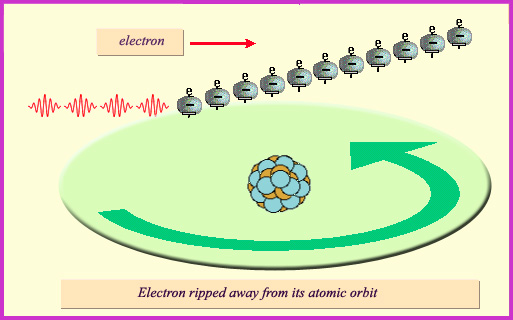
The photoelectric effect:
The photoelectric effect is the most common way for low-energy photons to interact with matter. It also takes place with gamma rays. A gamma collides with an electron orbiting an atom, expelling the electron tfrom its atomic orbit. The electron explusion is followed by a rearrangement of the atom’s electronic cohort to fill the gap. In the process the photon vanishes.
© IN2P3
Photoelectric effect : a photon is absorbed, an electron expelled from an atom.
The photoelectric effect is the most common form of interaction when the energy of the gamma rays is of the same order of magnitude as the energy binding atomic electrons to the nucleus. The gamma ray can then eject an electron away from an atom, sharing its energy between the electron and the excited atom. The gamma ray disappear, but the atom release later on it’s newly-acquired energy by emitting a ‘fluorescence’ photon, generally an X-ray. When an outermost electron replace an internal expelled electron, energy is released in the form of such fluorescence photons.
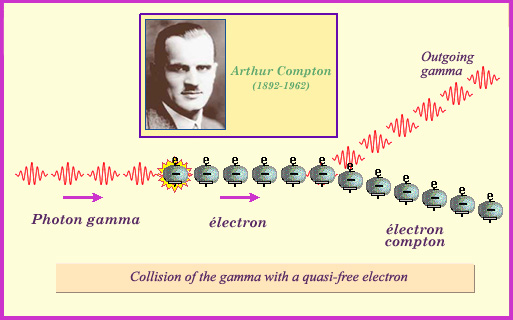
The Compton effect:
This second process is the most common way for gamma photons with mid-level energies to interact with matter. It resembles the photoelectric effect in that gamma rays collide with atomic electrons, but in addition to expelling the electron the photon is replaced by an outgoing photon with a lower energy. The Compton effect predominates when the incoming photon’s energy is far greater than that binding the colliding electron to its atom. The process gets its name from the American physicist Arthur Compton.
© IN2P3
Compton effect : elastic collision with an atomic electron
When the gamma photons have higher energies, the dominant interaction becomes the Compton effect, which is the collision between an incoming gamma ray and an electron. The initial energy is shared between the photon and the electron, which gradually slows down by ionising the atoms around it (much like a beta electron). The newly-formed gamma ray propagates in turn through the material until its it loses its energy by colliding with an other electron.
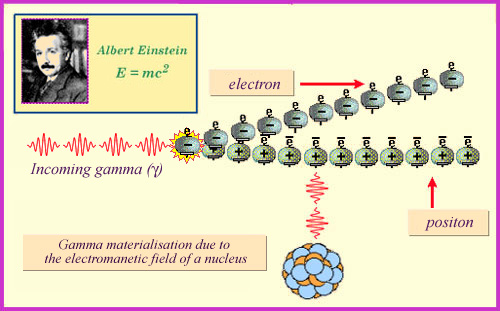
Pair Production
Pair Production is the production of a pair of mutual antiparticles, an electron and a positron. This process only takes place for photon energies above 1.02 MeV, the minimum energy required to create the two particles (according to the E=mc2 Einstein mass formula). The majority of gamma coming out of radioactive decay do not reach this threshold. Pair production occurs mainly with more energetic gamma produced by accelerators or found in cosmic rays.
© IN2P3
Electron-positron pair production :
When the energy of the gamma ray exceeds one million electronvolts, a third effect comes into play in which the photon interacts with a nucleus to produce an electron and a positron. The gamma ray energy is transformed into matter, with the strong electrical field of the nucleus acting as a catalyst. This pair production occurs more frequently than the Compton effect for high energies, but remains marginal in the field of radioactive phenomena.
Gamma attenuation :
The exponential attenuation of a narrow beam of gamma rays passing through an homogenous layer of material is characterized by an attenuation coefficient. The higher this coefficient, the faster is the attenuation. The attenuation coefficient is a measure of the interaction probability (called cross-section) of the gamma rays with the matter or the target. It is obtained by summing the contributions of the three effects: the photoelectric effect, the Compton effect, and (to a lesser degree) pair-production.
There is usually a significant probability that gamma rays travel a relatively great distance without being involved in any interaction. As a result, gamma radiation is both more penetrating and more difficult to stop than either alpha or beta. In laboratories and near accelerators, the custom is to use thick lead screens – a cheap and effective material – as gamma rays are more easily absorbed by heavier nuclei.
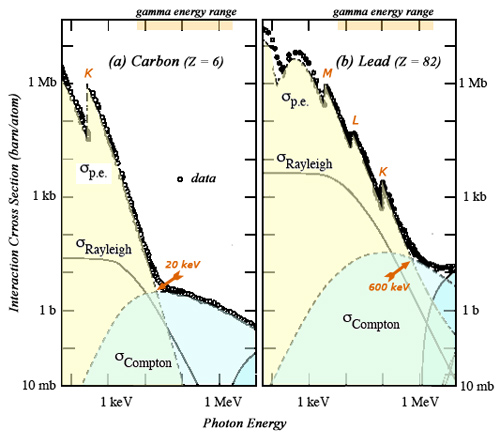
Competition between the Compton and photoelectric effects
The two main processes of gamma interaction with matter compete in the energy range of radioactivity gamma (from a few keV to 1 or 2 MeV). As can be seen on the examples of carbon and lead, the outcome of the competition depends on the gamma energy, the electron density and the atomic nucleus electric charge Z. The interaction probabilities for photoelectric effect are far higher from those of the Compton effect in the low energy range. Compton Effect becomes dominant for energies above 600 keV for lead and only 20 keV for carbon.
© IN2P3 (Source Particle Data Group)
The effects of gamma rays are much less localized thant those of alpha and beta rays. They are felt in larger volumes. This is why the gamma rays of cobalt-60 are used for example for food sterilization and disinfection of mummies. Low dose of gamma are used for medical diagnosis because they are less harmful in case of internal radiation. For this reason, technetium-99m, which is one of the few nuclei that emits only gamma, is highly prized.
NEXT : Photoelectric Effect,
NEXT : Compton Effect
Other articles on the subject « Radiations effects in matter »
Charged Particle Effects
A gradual loss and transfer of energy Alpha rays, fission products ; heavy, slow and ionizing par[...]
Alpha Rays in Matter
An atomic bulldozer, strongly ionizing along a very short path Alpha particles are simultaneously[...]
Beta Rays in Matter
Light electrons : a chaotic journey through matter Beta electrons and positrons have equal and op[...]
Bremsstrahlung
A relativistic phenomenon that applies to electrons and positrons… The phenomenon of bremss[...]
Neutral Particle Effects
Energy transfer by proxy… Neutral particles that are of interest in the field of radioactiv[...]
Cherenkov Effect
When an electron goes faster than light in air and water … The Cherenkov effect occurs when[...]
Cross Section
Cross section or the interaction probability of a particle Cross section is the name given by phy[...]
Photoelectric Effect
The most effective mechanism of photon absorption The photoelectric effect is the phenomenon that[...]
Compton Effect
Photons as projectiles and electrons as targets The Compton effect is the name given by physicist[...]
Macroscopic Effects
Effects on inert or organic matter The ionisation of atoms surrounding the trajectory of an alpha[...]