The fundamental constituents of matter
On the most elementary scale, the world around us is made up of four corpuscles. The first two are called the up and down quarks. Hidden in the nuclei of atoms, they are not familiar to us. The third is the electron, discovered more than 100 years ago. The fourth fundamental corpuscle is the neutrino, an electron that has lost its electrical charge. Although it is omnipresent in nature, the neutrino is very difficult to observe. Electrons and neutrinos are leptons.
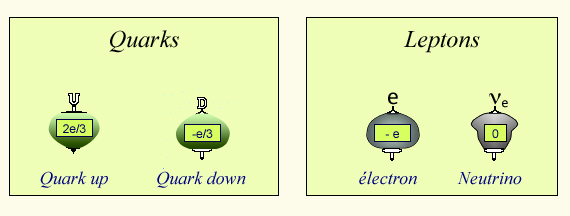
Quarks and Leptons
Nuclear matter consists of particles called quarks, belonging to two species called by the physicists « up » and « down ». A third elementary corpuscle is the electron present around the atoms. A fourth is the neutrino-electron, very difficult to observe, which can be considered as a neutral electron . They are represented here as small tops to figure their internal rotation. In the center, the values of their electric charge with respect to the elementary charge e of a proton.
© IN2P3
These four corpuscles are to this day, considered as elementary, without structure, punctual. For example, attempts to measure the size of an electron lead to a radius that would be more than ten thousand times smaller than that of a proton. Despite this tiny size, quarks, electrons and neutrinos have two states of rotation or spin. To recall, the four corpuscles were symbolized on the figure by small tops.
The electron was the first of these corpuscles to be discovered by JJ Thomson in 1896. The neutrino was proposed as a hypothesis by the Swiss physicist Wolfgang Pauli in 1930 but it was not until 1956 that the Reines and Cowan experiment demonstrates its existence. The existence of quarks, proposed by the American physicist Murray Gell-Mann, took hold in the 1970s through numerous experiments, in particular through the discovery of new species of quarks with very short lifetimes, that have vanished from our environment.
Quarks and leptons differ in how they interact. Three fundamental forces or interactions are present in the field of elementary corpuscles: Strong interactions responsible for nuclear forces in the nucleus, electromagnetic interactions that cause, for example, the rotation of electrons around the nucleus and weak interactions that manifest through beta radioactivity .
Quarks interact through these three forces. They are confined within protons and neutrons and more generally in nuclei by the very intense attraction resulting from the strong interactions. As a result, no free quarks have been observed to date. If one tries to eject a quark during a violent collision, it recombines immediately with other quarks or antiquarks produced in the collision. For example, because it was impossible to observe alone, physicists could not measure the mass of the quarks up and down. According to today theories, up and down quark masses would be small, of the order of a few masses of electrons.
The electron is insensitive to strong interactions and does not feel nuclear attraction. Because of this, he travels in the vast world. through its electrical charge, it orbits around the nuclei, emits and absorbs light waves and other photons. It provides carries electric currents when asked by humans.
As for the neutrino, it is insensitive to strong and electromagnetic interactions. It can only interact with weak interactions. It interacts so weakly that most neutrinos produced in the sun can cross the earth without any hindrance.
Corpuscles like quarks and leptons, which have two internal states of rotation (spin), are called fermions. They bear the name of the great Italian physicist Enrico Fermi who made a first theory of them around 1930. The fermions are subject to the exclusion principle, formulated by Wolfgang Pauli in 1925. The exclusion principle plays a very important role in atoms and nuclei. For instance, it forbids to two electrons with the same spin state to be close of each other. Without this principle, atomic electrons may crash on the nucleus, instead of turning around it and occupying a relatively large volume at the elementary scale. There would be neither atoms nor molecules !
Other articles on the subject « The atomic nucleus »
The proton
The charged constituent of the nucleus The nucleus of the hydrogen atom consists of one solitary [...]
The Neutron
The neutral partner of the proton The neutron is, along with the proton, one of the two constitue[...]
Isotopes
Nuclear variants of a given atom … Free of all electric charge and present only in the nucleus, n[...]
Nucleus Energy Levels
Analogies with the atomic energy levels … At first glance, nuclei seem to be very different[...]
Quarks and Gluons
How quarks interact within nuclei … To describe radioactivity and nuclear reactions such as[...]
Matter and Antimatter
Nature loves symmetry. However, one can not dream of a matter less symmetrical than the matter wh[...]