RBS or « Rutherford Backscattering Spectrometry »
The « Rutherford Backscattering Spectrometry” or RBS is a ion beam analysis technique widely used in materials science, and. thanks to the AGLAE accelerator also at the Musée du Louvre laboratory. This tool bears the name of the great new Zealander physicist Ernest Rutherford and is an application of the famous experience that enabled him in 1903 to demonstrate the existence of the atomic nucleus.
The beam particles, accelerated by AGLAE to an energy of several MeV , are directed onto the sample to be analyzed. The beam is composed of positive ions, either protons or helium nuclei (α particles). Sets of electromagnetic lenses focus the beam on the sample to a diameter of several microns. Because of this beam concentration, specialists talk of a nuclear microprobe
, are directed onto the sample to be analyzed. The beam is composed of positive ions, either protons or helium nuclei (α particles). Sets of electromagnetic lenses focus the beam on the sample to a diameter of several microns. Because of this beam concentration, specialists talk of a nuclear microprobe

Chandelier in the cathedral of Hildesheim in Lower Saxony
Hildesheim Cathedral is proud of a magnificent crown of light from the Ottonian period suspended in its choir. The Hezilo chandelier includes 12 towers and 12 gates linked by fragments of walls, depicting the heavenly Jerusalem. In the past, damaged parts were replaced with new pieces, gilded by electrolysis. In 2000-2001, two towers, one door and two fragments of wall have been sent to the C2RMF laboratory for gilding analysis before their restoration, using Rutherford backscattering with AGLAE.
© C2RMF
The RBS method consists in counting the number of incident projectiles ions that bounce back when they are repelled by the electric field of target nuclei in the sample. Detectors able to count the backscattered ions and mease their recoil energy are disposed at a diffusion angle, generally in the range 150-170 ° relative to the beam direction. Even with intensities low enough to avoid damaging the sample, the beams contain billions of ion projectiles (remembers the value of Avogadro number !). The detectors count in a short time thousands of backscattered ions.
For a given angle, the ion recoil energy is characteristic of the mass of the target nucleus. The heavier the nucleus, the higher isthe recoil energy and more important the probability of observing this backscattering because it varies as the square of the electric charge Z of the nucleus. The technique is suitable for the analysis of intermediate or heavy elements in a light material.
Like alpha particles, the accelerated ions have a very short range in matter, which depends on the acceleration energy. The analysis concerns only the superficial part of the material (a few microns or less) as the coating of a painting or a thin foil. The spectrum obtained with a thicker target has a particular form consisting of successive steps with an edge at an energy characteristic of each component and height in first approximation proportional to the concentration of the element.
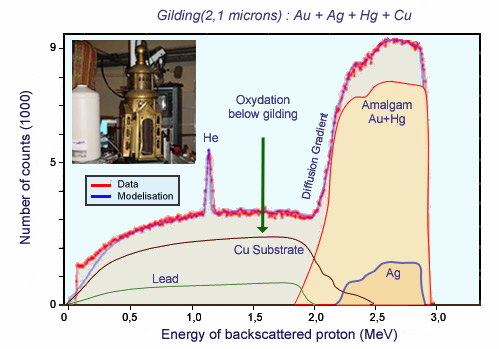
RBS spectrum of a Hildesheim chandelier gilding
RBS measurements of the composition and thickness of the layers of gilding in authentic parts (mercury amalgam) and parts restored (gold plating). Rutherford backscattering (RBS) provides access, from the surface, to the composition in chemical elements. It was shown that the authentic and restored gilding had the same composition, presumably to ensure color consistency. The gold titre was less than 21 carats, the thickness of the gilding was 2 to 5 microns for the mercury gilding and 0.2 to 0.5 microns for the gold electrolytic plating. These represent thick coatings, testimony of a special care in the treatment.
© LRMF
The RBS spectrum also provides information on the depth distribution of target components, due to the loss of energy of the incident ion in the incoming path and that of the ion diffused after going back. Physicists use tables to take into account these energy losses and interpret RBS spectra.
PIXE and RBS analyses are often combined. The PIXE technique is extremely sensitive. It gives access to the concentration of chemical elements heavier than sodium (Z> 10), even in trace amounts in the near-surface region. The simultaneous implementation of PIXE and RBS can provide the depth profile of major abd minor atomic constituents. One is able to measure local concentration of elements at the level of millionth of a gram per gram ( ppm).
Learn more :