Separate packages for minor actinides and caesium-strontium
As the change in the heat released by a standard vitrified package shows, the dominant elements in the first hundred years are two short lived-fission products, caesium-137 and strontium-90. The idea is to remove the caesium and strontium from the package destined for disposal, instead of removing the minor actinides for transmutation. The half-life of both the caesium and the strontium is around 30 years. The package would then be sent for disposal once these two elements had been removed.
Partitioning caesium and strontium is feasible. It was done at the time of the Fukushima accident, in which radioactive water was decontaminated by removing these two radioelements. Partitioning without transmutation would offer the possibility of adapting waste management to the type of radioactive waste and would open up interesting possibilities.

To give an example of the advantages offered by partitioning radioactive species, we have removed the fission products with a high thermal output (caesium and strontium) from the standard package so that they can be conditioned separately in a second package. The more compact package destined for disposal would contain mainly long-lived but not very mobile minor actinides. After 35 years it would release only the equivalent amount of heat to an electric light bulb.
IN2P3
After 35 years the compact package, containing the minor actinides, would release only one sixth of the heat of a standard package, the equivalent of an electric light bulb. The footprint of a disposal facility is dictated mainly by the amount of heat released by the high-level waste packages when they are sent for disposal, which means they have to be spaced out so that the host rock temperature does not exceed 100°C. If the packages gave off the equivalent of a modest electric light bulb, much more waste could be disposed of in a waste disposal facility like CIGEO.
What should be done with the partitioned caesium and strontium? The idea is that they would be incorporated into a second vitrified package. Initially this package would be hot (exothermic) and would have to go into long-term storage. But after 300 years, or 10 half-lives, its radioactivity and the amount of heat released would be 1000 times lower. It would only be giving off one watt: the exothermic package would have gone cold! After 600 years the strontium-90 would be one million times less radioactive, and so would the caesium-137. But by that time another fission product 76,000 times less active, caesium-135, would have become dominant.
At the outset the caesium-137 in the package would be very radioactive because of the powerful gamma rays it would emit. The gamma radiation coming out of the package could be reduced by encapsulating the glass package in lead, a very absorbent shield. The radiation from the strontium, which is not very penetrative, would be absorbed by the glass. A vitreous material like R7T7 glass can easily retain caesium and strontium atoms for several centuries.
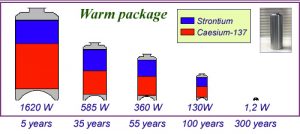
We have taken the fission products with a high thermal output (caesium and strontium) out of the standard package so that they can be conditioned separately in a second package. The exothermic package containing the strontium and caesium does not have to be put into a deep geological disposal facility at the start. It just needs to be left to wait for a long while. Because of radioactive decay, after 300 years, it would only be giving off one watt, since its radioactivity would have decreased by a factor of a thousand.
IN2P3
And now for a little science fiction… Due to radioactive decay (a factor of a thousand in 300 years) and good radiation protection, the package will have become harmless during its long-term storage. Experience with glass raised from Greek and Roman wrecks has shown that glass can withstand sea water for at least 2000 years. Our distant descendants will not have to be as afraid of the waste as us and can throw the harmless package into a trench on the seabed or bury it underground. Any rare gamma rays that come out of it would then be absorbed by a metre of water or soil. Audaces fortuna juvat (Fortune smiles on the brave), as the Ancient Romans used to say!
NB: The risk presented by the caesium-135 is negligible. Besides not being very radioactive, caesium-135 (which has a half-life of 2.3 million years) does not emit gamma rays. The low-energy beta rays it emits would be stopped by a few centimetres of water, glass or soil.