Cadmium and boron: two effective neutron absorbers
Both in radiation protection and in regulating reactor operation, it is necessary to be able to absorb or reduce the flux of neutrons.
Neutron-absorbing materials are used for this purpose, either in metallic form or as compounds. The main neutron absorbers are cadmium and boron. Hafnium is also used, and sometimes a rare earth element, gadolinium. These natural elements owe their absorbing properties to the presence of isotopes for which the probability of neutron capture is exceptionally high.
These absorbers are used in reactors because their capture probability—called the cross section—is much higher than that of the fissions that need to be regulated. They behave like poisons. Depending on the quantity introduced, their insertion slows down or suppresses the chain reaction.
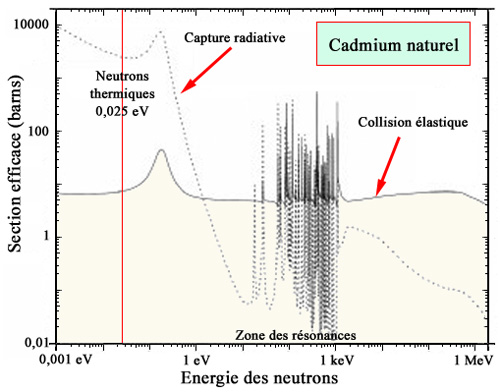
Absorption of neutrons by cadmium
Natural cadmium is composed of several isotopes, one of which, cadmium-113, is an effective neutron absorber. The curve of the probability of neutron capture by cadmium shows a giant resonance that peaks at nearly 10,000 barns for neutron energies on the order of 0.1–0.2 eV. This value is more than 10 times higher than the probability of fission of uranium-235 by thermal neutrons (0.025 eV).
© IN2P3
The main neutron absorber used in metallic form for control rods in nuclear reactors is cadmium. The cadmium isotope that acts as an absorber is cadmium-113. This isotope makes up 12% of natural cadmium. It has a very large cross section (20,600 barns), nearly a thousand times or more than that of the other isotopes.
This “giant resonance” of the capture cross section is located just above the range of thermal neutrons. It occurs when neutrons enter this energy range favorable to the fission of uranium-235 nuclei. The insertion of cadmium control rods is therefore particularly effective at slowing down or even stopping the chain reaction.
A good absorber should not generate radioactivity. The unstable isotopes that may be created by neutron capture (cadmium-115 and 117) have lifetimes on the order of an hour, leading to only marginal production of indium.
Gadolinium has two highly absorbing natural isotopes, gadolinium-155 (61,000 barns) and 157 (254,000 barns). Gadolinium-158 is stable. Its very low capture cross section (2 barns) makes subsequent captures and the resulting activation products negligible.
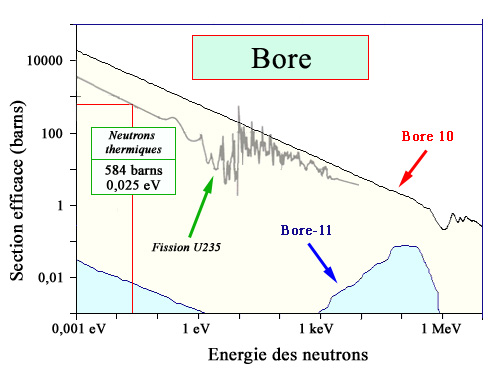
Absorbing power of boron
The probability (cross section) of fission of uranium-235 nuclei is compared with the probability of neutron absorption by the two isotopes of boron: boron-10, which makes up 20% of natural boron, and boron-11, which makes up 80%. The comparison shows that the absorption probability of boron-10 is much higher than that of fission for all neutron energies, unlike cadmium-113, for which the very high absorption probability was due to a giant resonance located below 1 electronvolt of energy.
© IN2P3
Boron is a light element, located between beryllium and carbon in the periodic table. Boron is an excellent neutron absorber. Natural boron consists of 19.9% boron-10, whose absorption cross section for thermal neutrons is 3,837 barns, and 80.1% boron-11, with a low cross section (5 barns). Most neutron captures therefore occur on boron-10. Normally this capture should result in boron-11, but more often it triggers an alpha decay: the nucleus splits into a lithium-7 nucleus and a helium nucleus. Neither is radioactive. The few neutron captures undergone by boron-11 transform it into boron-12 and then, in a fraction of a second, into stable carbon-12.
Boron is used, in the form of boric acid dissolved in the moderator water, to control reactors whose core has just been loaded with fresh fuel enriched in uranium-235. Later, when spent fuel is removed from the reactor to be cooled in a storage pool near the reactor and then in large storage pools at the reprocessing plant, boron is added to the water in these pools. The stored uranium still contains about 1% fissile uranium-235. Although the configuration is no longer that of a reactor, the introduction of boron dissolved in water prevents any unintended development of a nuclear chain reaction. As an additional precaution, spent fuel assemblies are placed in borated steel racks.
Boron was also added for the same reason after the Fukushima accident to the water injected into the vessels of the damaged reactors, as well as into the adjacent storage pools. The addition of this boron, combined with the presence of cadmium from the control rods remaining in the vessels, prevents any risk of a nuclear explosion.
Boron also plays a role as a radiation shield and in neutron detectors.