Radioactivity, a tool for knowledge
Radioactive sources allowed Ernest Rutherford, Marie Curie and others to conduct the first examinations of the nucleus. In the following century, radioactivity has revealed itself capable of a myriad of applications, and has become an invaluable source of subatomic information. The very nature of radioactivity allows scientists to trace atoms or molecules with great sensitivity.
The large diversity of radioactive elements together with the tremendous spectrum of their respective half-lives leads to many applications. Some can survive for billions of years, while others disappear within minutes. This range, from the quasi-permanent to the instantaneous, allows for radioactivity to be used in practically every sphere of human endeavour.
In biological research, short-lived radioactive tracers such as tritium or phosphorus 32 are universally popular. A radiotracer such as iodine 123 allows for the examination of thyroid problems with a gamma camera.
Carbon 14, with its half-life of 5,700 years, has revolutionised the field of archaeological dating. Using the mathematical laws underlying all radioactive decay, objects and places up to 40,000 years old can be accurately dated. Other radioactive techniques (such as examining the ratio between quantities of potassium and argon present in a sample) can be used to date much older rocks.
Radioactivity also has uses in earth science and the study of the environment. It was in these fields that uranium, potassium and rubidium allowed for an accurate estimate of the age of the Earth. Climatologists measure solar activity by seeing how much beryllium 10 produced by cosmic rays gets absorbed by the polar ice caps. Oceanographers can, through measuring the abundance of carbon 14 in the oceans, retrace the flows of ancient streams.
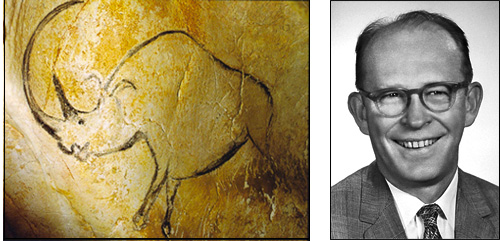
Rhino drawing of the Chauvet grotto dated by the radiocarbon technique. The American physicist and chemist Willard Libby (right) earned the Nobel Prize in 1960 for this technique.
© Grotte Chauvet
The discovery of radioactivity led to that of the atom nucleus and to the development of nuclear physics. This first step in the exploration of the infinitely small led to the birth of a new discipline, particle physics thanks to large accelerators. The CERN Courrier describes the history in France of these two disciplines in an excellent article : The rise of particle physics in France.
ACCESS TO CHAPTERS : SUMMARY
– Methods of detection : Extraordinarily sensitive detectors
– Radioactive dating techniques : Carbon 14, the age of the Earth
– Radioactive markers and tracers : Track down Nature with infinitesimal amounts of radioactive atoms
– Applications to earth sciences : Using radioactive atoms in Oceanography, climatology, etc ..